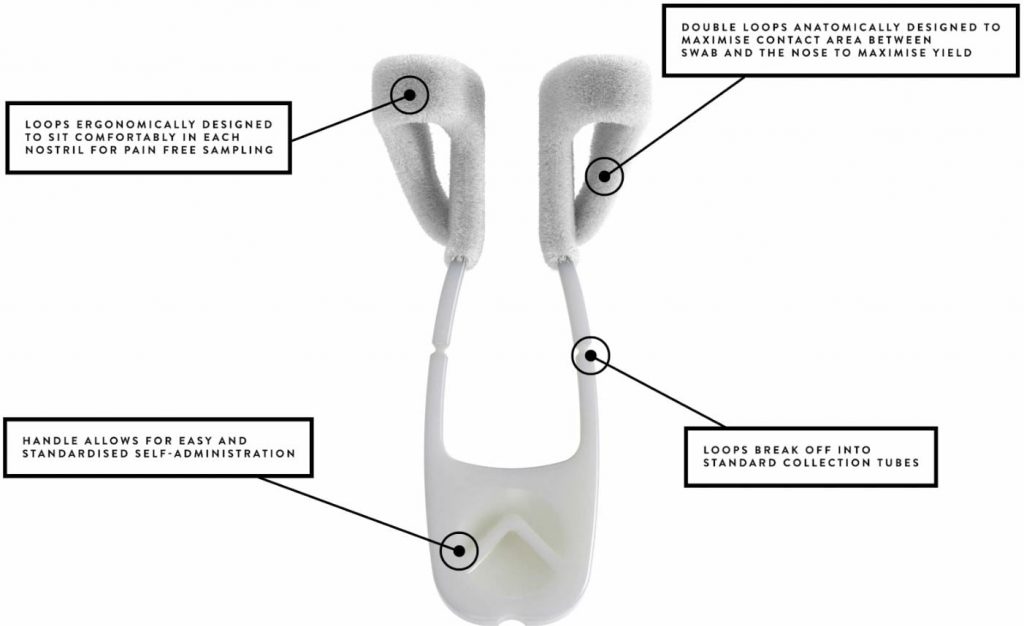
Rhinoswab Junior has been designed to deliver the comfort, reliability and performance of Rhinoswab enhanced by novel features that reduce the fear, anxiety and trauma associated with the use of traditional nasal swabs for children 4-12 years of age.
See how easy and comfortable this self-administered nasal swab works.
RHINOSWAB an easier, more comfortable way to swab.
Rhinoswab is a patented comfortable, self-administered nasal swab that provides high-yield sampling and fits existing pathology workflows.
The RHINOSWAB Advantage:
- Comfortable and pain free
- Self administered
- Dual Nosetril application for high yield
- Safer for healthcare workers
- Improved standardization of collection
- Works with both laboratory and PCR tests
See how easy and comfortable this self-administered nasal swab works.
Download flyer Junior Rhinoswab
TECHLAB® manufactures a variety of in vitro diagnostics for enteric diseases to help determine whether a patient has a disease caused by Helicobacter pylori, Clostridium difficile, protozoan parasites, foodborne illness, or IBD/IBS.
From mid September all TECHLAB® products are distributed bioTRADING in The Netherlands, Belgium and Luxembourg. Please visit our webshop or contact bioTRADING for product and order information.
MSC-96000 is a flexible Flocked Sampling Swab used for cell & virus specimen collection, and it has been designed for Nasopharyngeal clinical diagnostics.
It has been well recognized and adopted by the diagnostic test kit manufacturers who produce reagents in molecular genetics, forensics and
clinical laboratories sectors.
The perpendicular nylon fibers act like a soft brush, allowing the improved collection and release of both cellular and liquid samples.

Please contact us if you need a specific product, we are meeting many customers’ unique requests.
Rapid in vitro diagnostic test for the qualitative detection of SARS-CoV-2 antigen in nasopharyngeal samples.
Instructions for use
Features:
- Fast: get results in 15 minutes.
- Mass screening: Detection of SARS-CoV-2 proteins in nasopharyngeal secretions.
- Simple & affordable: An easy, simple and cheaper initial sorting of patients.
Please contact us if you need a specific product, we are meeting many customers’ unique requests.
CHROMagar™ Candida Plus
For detection and differentiation of major clinical Candida species, including C. auris.
CHROMagar™ have modified the formulation of their CHROMagar™ Candida so to have Candida auris growing in a very specific morphology: light blue colonies with halo and blue from the back of the plate.

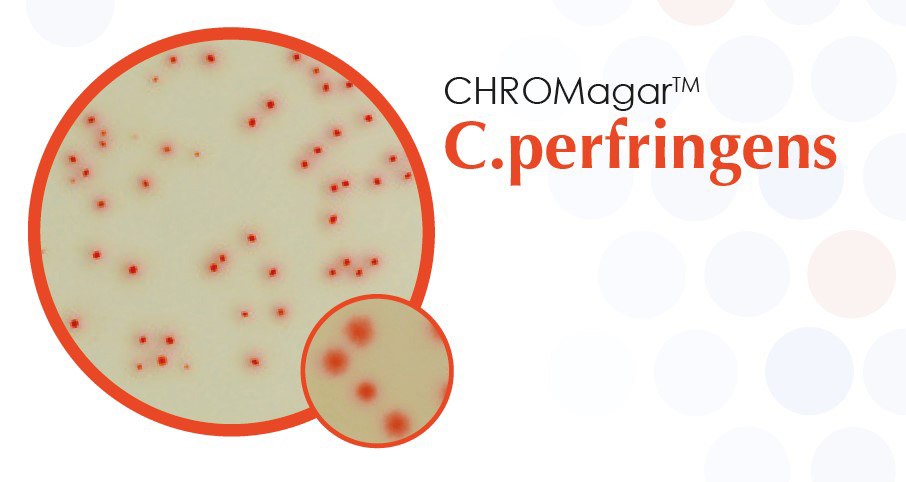
CHROMagar™ C.perfringens
For detection and enumeration of C. perfringens.
CHROMagar™ C.perfringens is the only culture media designed to detect and enumerate Clostridium perfringens in food and water samples. Specific and selective, this medium detects Clostridium perfringens colonies by an intense orange colouration, the other microorganisms being blue, metallic blue or inhibited.
Please contact us if you need a specific product, we are meeting many customers’ unique requests.
MSC-93050 is a flocked swab of sampling for Laryngopharyngeal clinical diagnostic, it utilizes state of the art “spray on technology” that the flocking process by means of an electro-static charge perpendicularly attaches millions of nylon microfibers on the medical grade handle tip.
The flocked swab is ideal for collecting large amount of cells and rapid elution of the specimens that instantly releases the cells into the transport medium. It has been well recognized and adopted by the diagnostic test kit manufacturers who produce reagents in molecular genetics, forensics and clinical laboratories sectors.

Please contact us if you need a specific product, we are meeting many customers’ unique requests.
The last concept added to the ALCOA+ principle is Data Availability.
This point is a consequence of the previous one (Data Endurance), i.e. data must not only exist, it must also be accessible whenever needed, during the whole data lifecycle.
Availability ensures data meets its use, since it can be used when needed. Data should be accessible at any time during the required lifetime. This also applies to any associated metadata. Additionally, data must be made available, securely, for the people who need it. Undoubtedly, the most efficient way to achieve the availability principle is recording data electronically.
Let’s look at an example of an FDA warning letter to better explain how the Availability concept should be applied to pharmaceutical companies:
FDA Warning Letter 320-19-16 (March 2019)
Your firm failed to ensure that laboratory records included complete data derived from all tests necessary to ensure compliance with established specifications and standards (21 CFR 211.194(a)).
You do not retain laboratory data generated by your analysts. During our inspection, your staff told our investigator that your firm’s laboratory worksheets contain only final laboratory results. You do not retain records of sample weights written on paper. The papers are discarded after your staff performs calculations.
Due to your failure to retain laboratory data, our investigator was unable to confirm the validity of your results. You could not provide any rationale for failing to maintain the complete data. Your staff further stated to our investigator that if a sample preparation is not good, it is thrown away and restarted. The staff does not retain that data.
In response to this letter, provide:
- A management strategy for your firm that includes the details of your global corrective action and preventive action plan. The detailed corrective action plan should describe how you intend to ensure the reliability and completeness of all data generated by your firm including microbiological and analytical data, manufacturing records, and all data submitted to FDA.
SherpaPharma offers full data Availability, high security and traceability.
The system is accessible over the Internet. Any approved user, with the right profile, can access the system from any location. Using date filters, and combining with any other filtering criteria (for example, manufacturing area, operators, sampling conditions, microorganisms identified, etc.), users instantaneously access the necessary sample data, including all the related information. For instance, in case of an OOS or an investigation, users can quickly check if the incidence is new or if it has been repeated under the same or different conditions. CAPA plans can use SherpaPharma to confirm if actions are effective.

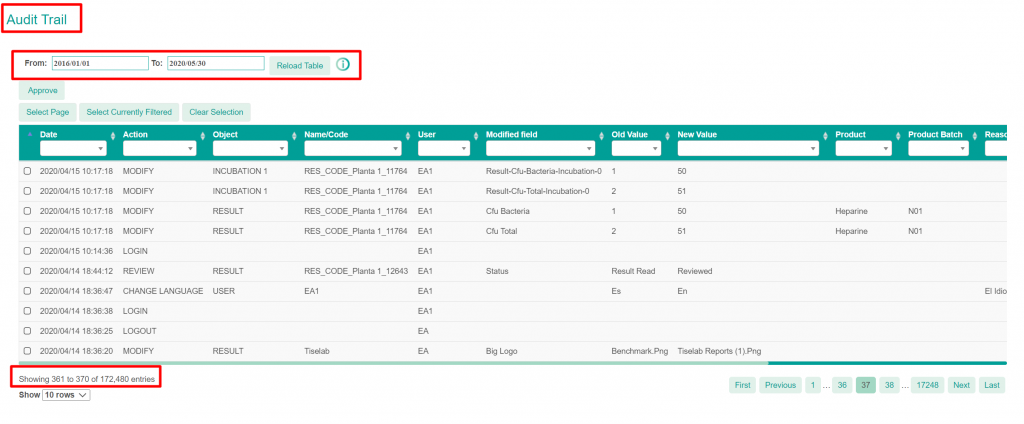
In addition, all data is securely stored in a data base that is daily backed up to a different geographical location. The system is monitored continuously and upgraded periodically to make sure that data will be available always and without occupying space in your location. SherpaPharma users do not need any more a warehouse for GMP microbiological documentation or local servers for copies of environmental data and metadata.
The COVID-19 pandemic has strained every aspect of healthcare, including Women’s Health & STI testing. Diagnostic labs around the world have struggled with shortages, shipping restrictions, and many other challenges, just to provide the testing that remains as critical as ever. To help labs stock these critical controls, we are offering exclusive savings on Women’s Health and STI Controls.
With the purchase of any of the Women’s Health and STI Controls, we are offering 2 of the following controls for free:
MBL8234 – Multi-Strain Human Papillomavirus (HPV) Control Panel (Inactivated Pellet)
MBL8235 – Multi-Strain Human Papillomavirus (HPV) Verification Panel (Inactivated Pellet)
MBL8240 – Mycoplasma genitalium Control Panel (Inactivated Pellet)
Place an order before September 1st to take advantage of these savings. Mention promo code three4oneQC to receive your discount.
CARBA 5 is a rapid lateral flow test that detects the 5 most prevalent resistance enzymes against carbapenems in one single test-strip. Ease in use and results after 15 minutes from colony. A phenotypic test with a 100% specificity, 100% sensitivity validated and tested by multiple national reference labs.
Detection limits:
– KPC 600pg/mL
– OXA 300pg/mL
– VIM 300pg/mL
– IMP 200pg/mL
– NDM 150pg/mL
CARBA 5 Flyer
Instructions for use
ASM poster 2017 CARBA 5
Please contact us if you need a specific product, we are meeting many customers’ unique requests.
Go to webshop