
AnalytiChem Netherlands is preparing to implement a new ERP system, SAP S4/HANA, as part of a broader effort to strengthen business processes, improve service reliability, and enhance collaboration with customers and partners.
The go-live is planned for the first two weeks of January 2026 and represents a significant step forward in operational efficiency and digital transformation.
As part of the project plan, regular checkpoints have been established to closely monitor the progress of the implementation. Internal teams are working across departments to ensure alignment and minimize any disruption during the transition.
What to Expect During the Transition
Despite the extensive preparation, short-term impacts may occur during the initial transition phase, including:
- Delays in order processing or confirmation
- Changes in invoice formatting or reference numbers
- Adjustments to delivery timelines
Dedicated teams are in place to manage and monitor the process, helping to maintain high service levels throughout the change.
Supporting a Smooth Transition
To reduce risk and ensure continuity of supply during this change, the team encourages:
- Placing orders in advance for products needed in early January
- Including clear order references (e.g. PO numbers, delivery dates) to facilitate processing
- Maintaining close communication with the regular sales or customer service contact
A step forward in process excellence
The move to SAP S4/HANA will deliver long-term benefits for both AnalytiChem and its customers, including more streamlined operations, greater data consistency, and improved responsiveness.
Updates will continue to be shared as the go-live date approaches.
We are delighted to share that AnalytiChem has globally launched Redipor® by AnalytiChem, a consolidated brand of ready-to-use culture media, integrating the legacy and expertise of three established brands: BioTrading, Redipor by Cherwell, and NEL. Together, we bring more than 150 years of combined expertise in high-quality, ready-to-use culture media to microbiologists and quality assurance professionals worldwide. Now unified under the AnalytiChem umbrella, Redipor stands as a symbol of trusted performance, innovation, and deep industry knowledge.
Redipor’s culture media products are crafted to meet the rigorous standards and regulatory demands of microbiologists working in critical sectors including Pharmaceuticals, Food & Beverage, Environmental and Healthcare. The product line includes a broad portfolio of media formats such as plates, bottles, tubes, and vials. These offerings are not only comprehensive but also customisable, allowing customers to request tailored solutions to fit specific applications and compliance requirements.
Read the full press release on the AnalytiChem website to learn more about how Redipor by AnalytiChem is delivering ready-to-use culture media with global reach and local expertise.
You may have seen that the new logo is already being used on our product labelling and documentation, such as Certificates of Analysis. The new Redipor logo will gradually replace the current logo on all related packaging, as existing stock is depleted. Until then, you may receive products with a combination of the new and old logo.
We are currently working on updating our website, images and literature with the new logo and are aiming for the full transition to be completed within 12 months after the official launch of 18th April 2025.
For faecal concentration of Helminth ova and larvae/Protozoa cysts and oocysts.
CE marked IVDR-conform.
Health and safety benefit
- Totally enclosed/sealed process
- Disposable device
- Single use, eliminates cross-contamination
- Pre-filled systems available
Performance benefits
- Optimum sample recovery
- Enhanced sample clarity
- Rapid four-step process
- Human resources optimised
- Reduces human error
Apafix™ Patented Universal Fixative
- Simplifies specimen processing.
- Formalin free fixative
- No warning labels – Non-toxic, Non-flammable, Eco-friendly
- Suitable for all Parasitological fixing
- Preserves morphology even with delicate organisms
- Compatible with PCR and other molecular methods
- Can be used in conjunction with EIAs.
- Suitable for use with a variety of common staining techniques
- Available in transport vial and Parasep® filled vials
Go to our webshop to order the products. The reference numbers you need to order are:
APA108801 Mini Parasep® SF 3.3ml Apafix™, 40 tests
APA145002 Midi Parasep® 6ml Apafix™, 40 tests
APA145003 Midi Parasep® 6ml Apafix™ + 80ml Ethyl Acetate, 40 tests
APA146003 Mini Parasep® 2.4ml Apafix™, 40 tests
APA146004 Mini Parasep® 2.4ml Apafix™ + 40ml Ethyl Acetate, 40 tests
APA149001 Midi Parasep® SF 8ml Apafix™, 40 tests
APA160001 30ml Transport Vial with 15ml Apafix™, 40 tests
Please contact us if you need more information. You can also find more information in the brochure.
TB is a serious disease that affects a significant portion of the world’s population. Diagnosis of TB is important not only for correct treatment of the disease but also for containment of the highly contagious infected individual.
MycoDDR is a reliable processing system that facilitates the digestion and decontamination of patient specimens in a controlled process, which is essential for mycobacterium survival and recovery. It empowers labs to maximize the retention of viable Mycobacteria via rigid pH control throughout the digestion/decontamination process.
This rigid pH control leads to faster culture results, improved smear quality, and decreases lab tech variability.
Benefits:
- Smears are easier to interpret
- Obtain culture results 1 – 7 days faster
- Reduce the risk of false negatives
- Ragid pH control will lead to more real positives
- Fewer specimens to reprocess
- Reduced risk of cross-contamination
- Easy to process specimens containing blood
- Eliminates user variability, reducing the chances for technical error and allowing samples to be easily processed
- Visual result confirmation
- Contain a pH indicator
Go to our webshop to order the products. The reference numbers you need to order are:
TBNN1010-2.5 (120 tests) (2,5% NaOH with NALC)
TBNN1010-3.0 (120 tests) (3% NaOH with NALC)
TBPN67-60 (60 tests) (neutralization)
TBRB30-60 (60 tests) (resuspension)
To have a proper number of reagents, order two of the neutralization and resuspension buffers for every TBNN1010 that is ordered.
Please contact us if you need more information. You can also find more information in the brochure and Package Insert.
NG-Test® CARBA-5 is a rapid in vitro diagnostic assay, for professional use only. It is a visual multiplex immunochromatographic (lateral flow qualitative assay for the detection and differentiation of the five most common carbapenemase families (KPC, OXA-48-like, VIM, IMP and NDM) from Carbapenem non susceptible pure bacterial colonies of Enterobacterales (including Escherichia coli and Klebsiella pneumoniae) and Pseudomonas aeruginosa in healthcare settings.
– Results within 15 minutes
– Provides information to inform appropriate antimicrobial therapeutic choices
– Support prompt infection control decision making and interventions
– Easily implemented into any laboratory settings with minimal training
– Minimal hands-on time
– Simple preparation protocol
– No instrumentation is required
– Can be performed directly from positive blood culture samples
– Sensitivity: 100% / Specificity: 100%
NG-Test® CARBA-5 detects the following variants:
– Type NDM: NDM-1 -2 -3 -4 -5 -6 -7 -8 -9 -11 -19
– Type KPC: KPC-1 -2 -3 -4 -5 -6 -7 -12 -14 -23 -28 -39
– Type IMP: IMP-1 -2 -4 -5 -6 -7 -8 -10 -11 -13 -14 -15 -16 -18 -19 -22 -26 -29 -31 -37 -39 -46 -47 -56
-58 -61 -63 -71 -79
– Type VIM: VIM-1 -2 -4 -5 -6 -19 -23 -26 -27 -31 -39 -46 -51 -52 -54 -56 -58 -59
– OXA-48-like: OXA-48 -162 -181 -204 -232 -244 -245 -370 -436 -484 -515 -517 -519 -535 -793
Please contact us if you need more information. You can also find more information in the brochure and instruction for use.
For orderering go to webshop.
This product can be used for the detection and isolation of Salmonella species. Often present in meat, eggs and dairy products, Salmonella spp are also found in water, with the serotypes S.typhi and S.paratyphi being responsible for typhoid and paratyphoid fever.
Salmonella surveillance represents the most common routine analysis in food chain processes. Consequently, improving the efficiency of the test will lead not only to a reduction in the number of contaminated foodstuffs on the market, but also to substantial economic savings in costs related to microbial quality control.
Salmonella is generally considered a non lactose-fermenting organism, but the recent revision of ISO 6579 for Salmonella testing is a result of the growing incidence of lactose positive Salmonella spp. isolated from cases of food poisoning.
Medium Performance
1. Meets ISO ISO 6579-1 requirements by detecting lactose positive Salmonella.
2. Easy to read with the naked eye: Another feature of this new media resides in its nice colour contrast due to the fact that E.coli are colourless. E.coli , which are usually present in abundance in samples tested for Salmonella and can potentially hide suspect colonies, are no longer a concern.
3. High Sensitivity and Specificity: Salmonella including S.typhi, S.paratyphi A and lactose positive Salmonella) → 99%*
*Specificity and sensitivity from scientific study: “Evaluation of a new chromogenic medium CHROMagar Salmonella Plus for the detection of Salmonella species including lactose positive Salmonella, S.Typhi and S.Paratyphi” de Beaumont C., Breuil J., Dedicova D., Tran Q. 2006. Poster presented during ECCMID meeting.
Please contact us if you want to receive more information about this product or look into the brochure where you also can find additional information.

• Evaluates the lab’s ability to analyze sample quality
• High quality, fit for purpose materials
• Supports evaluation of a lab’s competence to perform compendial tests
• Unbiased assessment of critical and measurable properties of analytical testing
• A variety of schemes for both quantitative and qualitative test performance
The Proficiency Testing Program aims to confirm the competency of an individual’s laboratory staff performance of conducting testing and analysis to ensure valid and reliable testing results and the overall proficiency of testing laboratories.
The USP Proficiency Testing Program is a strategic alliance between USP and NSI Lab Solutions to develop and market proficiency test schemes. NSI is a highly reputable player in the proficiency testing market with multiple accreditations: ISO/IEC 17043, ISO 17034, ISO/IEC 17025, ISO 9001. They have developed a dynamic set of schemes that evaluate a wide range of quality control analytical testing. The program provides an increased awareness of individual performance, supports accreditation requirements, strengthens quality management systems (QMS), builds confidence within multiple industries, and builds regulator assuredness during the registration and lifecycle of products.
This program offers participants an external unbiased quality assessment of proficiency that verifies the overall performance of the testing laboratory against other comparable testing laboratories. The Proficiency Testing Program leverages NSI’s broad experience to bring customers a diverse set of schemes.
See attached brochure for the proficiency Tests that are available from NSI Lab solutions.
Please contact us if you need more information

Custom Controls from Environmental Isolates. Your strain. Your format. Your convenience.
Pharmaceutical microbiologists around the world trust Microbiologics quality control microorganisms for Growth Promotion Testing (GPT) and other tests. Did you know that Microbiologics also makes custom controls from environmental isolates?
Microbiologics can produce controls from your environmental isolates that are ready to use. These custom controls offer many advantages over maintaining your own stock cultures for quality control:
- No need for freezing
- Fast dissolving pellet
- Pre-enumerated for your test methods
Microbiologics is committed to helping pharmaceutical companies protect their brand identities. We keep all organism information strictly confidential.
The below resources show why Microbiologics custom controls are the best way to demonstrate the effectiveness of your environmental monitoring program:
- Culture Collection Strains
- Example Environmental Isolates
- Custom Controls Brochure
- Pharmaceutical Microbiology Controls Brochure
Your Microbiologics Distributor
As your Microbiologics distributor, we can help you protect your consumers and your brand identity with custom controls from your environmental isolates.
Please contact us if you need a specific product, we are meeting many customers’ unique requests.
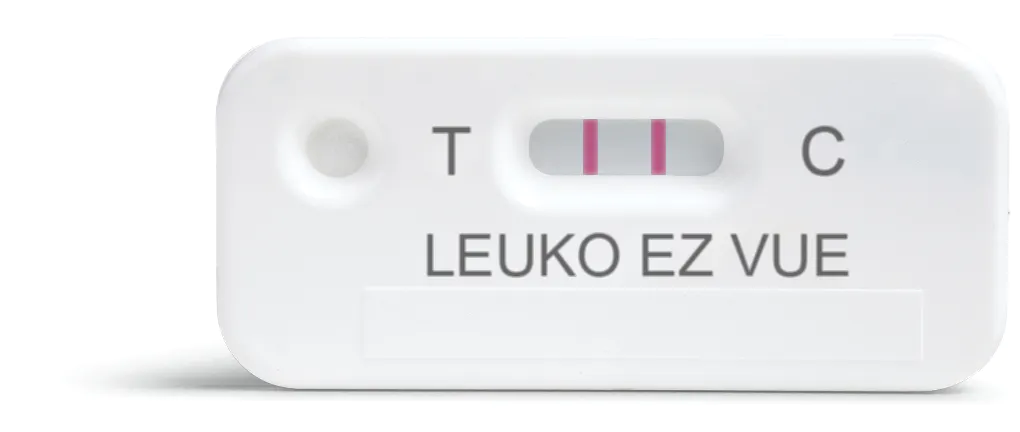
The LEUKO EZ VUE test is a qualitative assay which indications of the presence of leukocytes in fecal samples. It detects the presence of lactoferrin, a biomarker for intestinal inflammation, in stool samples.
Traditionally, intestinal inflammation has been identified through fecal leukocyte smear microscopy (also known as white blood cell smears). Traditional microscopy is a laborious process and suffers from false negative results as fecal leukocytes are unstable and begin to degrade immediately upon passage. Cellular degradation does not affect the results of the LEUKO EZ VUE test and offers results in a simple, 10 minute lateral flow format.
• Detects elevated levels of fecal lactoferrin, a marker of fecal leukocytes and an indicator of intestinal inflammation
• Simpler and more accurate than microscopy for fecal leukocytes
• Rapid results in 10 minutes
Simpler and more accurate!
Please contact us if you want to receive more information about this product. You can also find more information in the brochure.
The Universal Operator Broth Transfer Validation kit is a test kit developed to assess an operators ability to maintain the sterility of materials during the preparation of asepticaly prepared injectable dose forms.
bioTRADING offers all the products you need to easily asses the risk of microbial contamination of your Compounded Sterile Preparations (CSPs). Our products are designed to help you follow PIC/S or USP<797> guidlines.